QUALITY MANAGEMENT SYSTEM
General
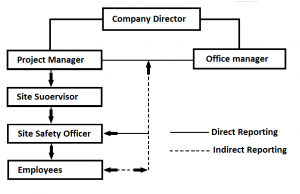
Management responsibility is concerned with operational obligations, through the allocation of the necessary resources (resources management), resulting in a quality product (product realization) and ultimately satisfying the client’s needs. To ensure a continuous improvement process provision has been made for measurement analysis and improvement, this is a quality control mechanism.
Documentation Requirements
Document and data control policies and procedures exist to ensure the right people get the right documents and information to ensure high quality performance. These policies also exist to ensure no data is lost or missed.
Control of Documents